Using neurobiology to understand and cure disease.
I’m currently available for full-time hire and consulting engagements.

About Logan
I am a neuroscientist interested in understanding fundamental neurobiology to cure disease and build better models of the brain. I completed my doctorate at Purdue University, where I developed a zebrafish-based drug-screening platform to identify compounds for treating retinal degenerative diseases. Currently, I am completing postdoctoral studies at UC Berkeley. I am investigating the impact of maladaptive physiological changes to retinal neurons following photoreceptor degeneration.
I have over 10 years of experience in translational neuroscience, advancing scientific discoveries from the laboratory to clinical application. I strongly believe that insights from molecular and cellular neurobiology are essential for informing the development of new treatments and accurate neural models. To this end, I design my studies using a bottom-up approach, progressing from molecular mechanisms to neural circuitry and ultimately to behavior.
I am committed to training the next generation of neuroscientists. Throughout my career, I have provided one-on-one mentorship to over 20 undergraduate students in the laboratory, helping them develop their knowledge and technical skills. My mentees have gone on to pursue their intended career paths, including admission to medical school and graduate programs in neuroscience.
Tool Development
New questions demand new tools.

A twisted bipolar electrode fabricated from insulated Nichrome wire. Designed to flank the optic tract in ex vivo brain slice preparations for electrical stimulation. Tip-to-tip is approximately 50 microns.
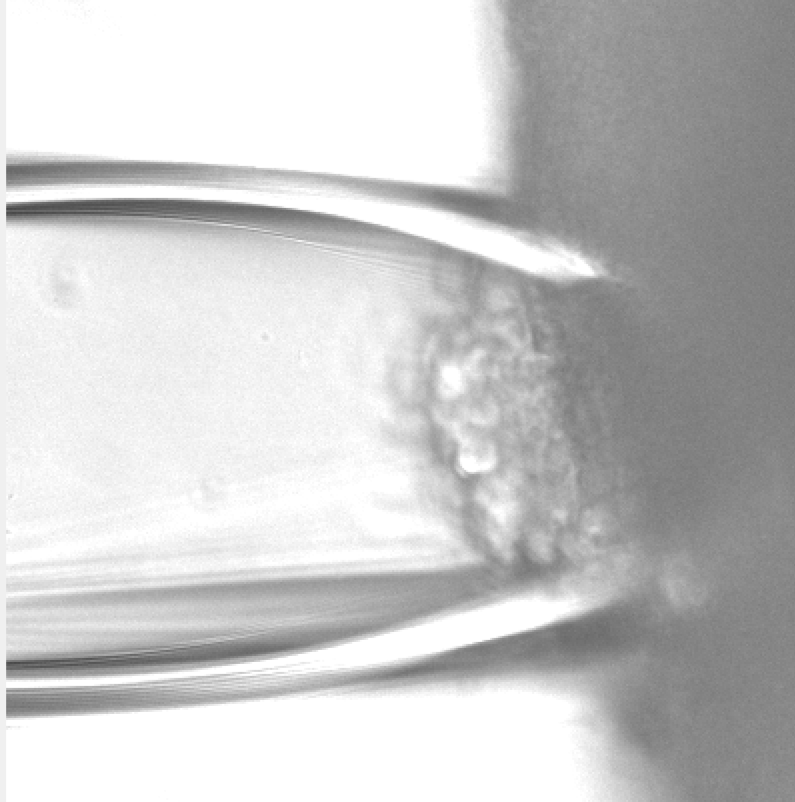
A custom glass suction electrode attached to the side of a brain organoid. This allows for the electrical stimulation of neurons to record calcium influx and fluorescently detect neurotransmitter release.

A piece of retina mounted onto a multielectrode array (MEA). Dialysis tubing is glued to a piece of silver metal to create an ion-permeable anchor for improved contact with the electrodes.
Functional Microscopy
Seeing is believing.
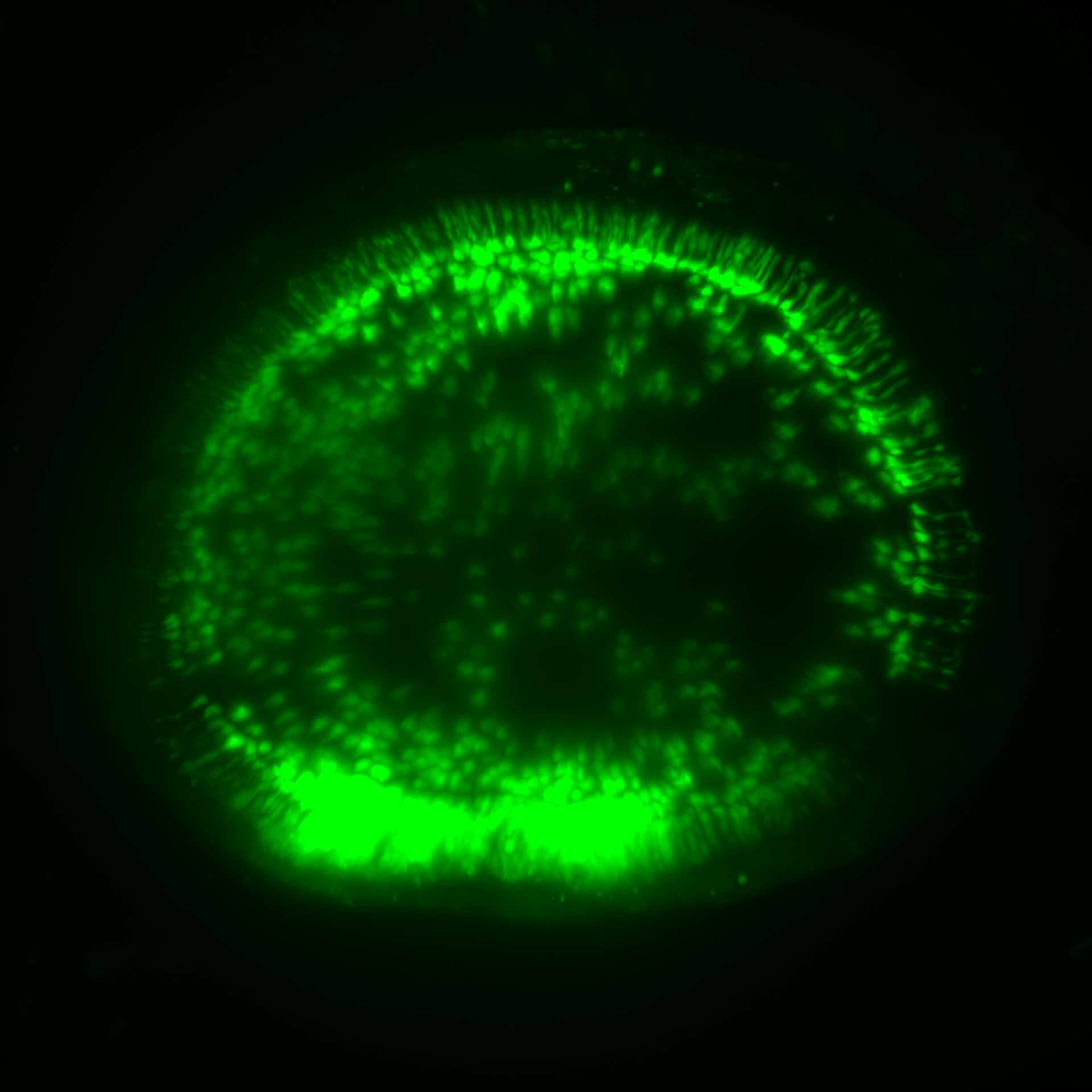
Glutamate release from a living flat-mount retina within the On-plexiform layer. Trans-retinal stimulation depolarizes retinal bipolar cells, evoking glutamate release onto postsynaptic retinal ganglion cells expressing iGluSnFR.
Glutamate release throughout the entire inner plexiform layer with retinal trans-stimulation visualized with iGluSnFR. Optical sections are reconstructed to investigate differences in glutamate release between Off-plexiform layers (top half) and On-plexiform layers (bottom half)
Calcium imaging of neurons within a brain organoid. A glass suction electrode is attached to the organoid, and a current is passed through to depolarize neurons to trigger voltage-dependent calcium influx.
Calcium imaging of the AIY interneuron in a freely awake and behaving Caenorhabditis elegans worm on an agar slab with a temperature gradient. The neurite of the AIY cell receives synaptic input from the AFD temperature-sensing neuron, driving calcium influx when the worm moves in a warm direction (outer ring of the inset). The soma of AIY exhibits oscillating calcium signals as the head of the worm deflects dorsally and ventrally, encoding proprioception. This was completed at the 2018 Neural Systems and Behavior Summer course at the Marine Biological Laboratory at Woods Hole with the Colón-Ramos Lab.
Electrophysiology
Understanding the fundamental membrane properties of neurons is vital for understanding neural systems.

An acute brain slice preparation with the optic tract left intact to the visual thalamus. A bipolar electrode is positioned in the ventral brain flanking the fiber tract (dark grey band). Electrical stimulation of the optic tract evokes synaptic release from presynaptic retinal ganglion cells to thalamocortical relay cells in the dorsal lateral geniculate nucleus (dLGN).

A dye-filled AII amacrine cell in the mouse retina. This neuron receives input from the rod bipolar cell and carries it into the cone pathway through gap junctions. Voltage-gated sodium channels amplify this signal, allowing for vision in dim light and detection of spatially correlated stimuli.

A three-electrode configuration of a Scientifica SliceScope for multi-site stimulation and recording. This is a versatile platform for performing electrophysiology experiments, such as patch-clamp.
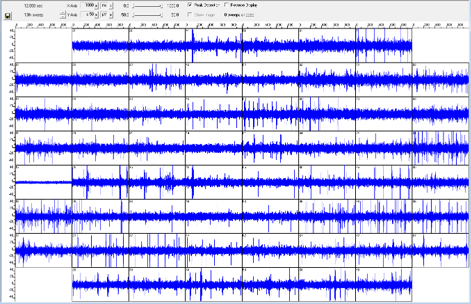
A recording from a multielectrode array (MEA) from Multichannel Systems. A retina undergoing photoreceptor degeneration has been mounted on the array so that retinal ganglion cells are in contact with the electrodes. In this retina, the retinal ganglion cells are spontaneously hyperactive, firing without any upstream light information. Straight lines in the channels are action potentials. This technology allows for high-throughput electrophysiological recordings of neurons.
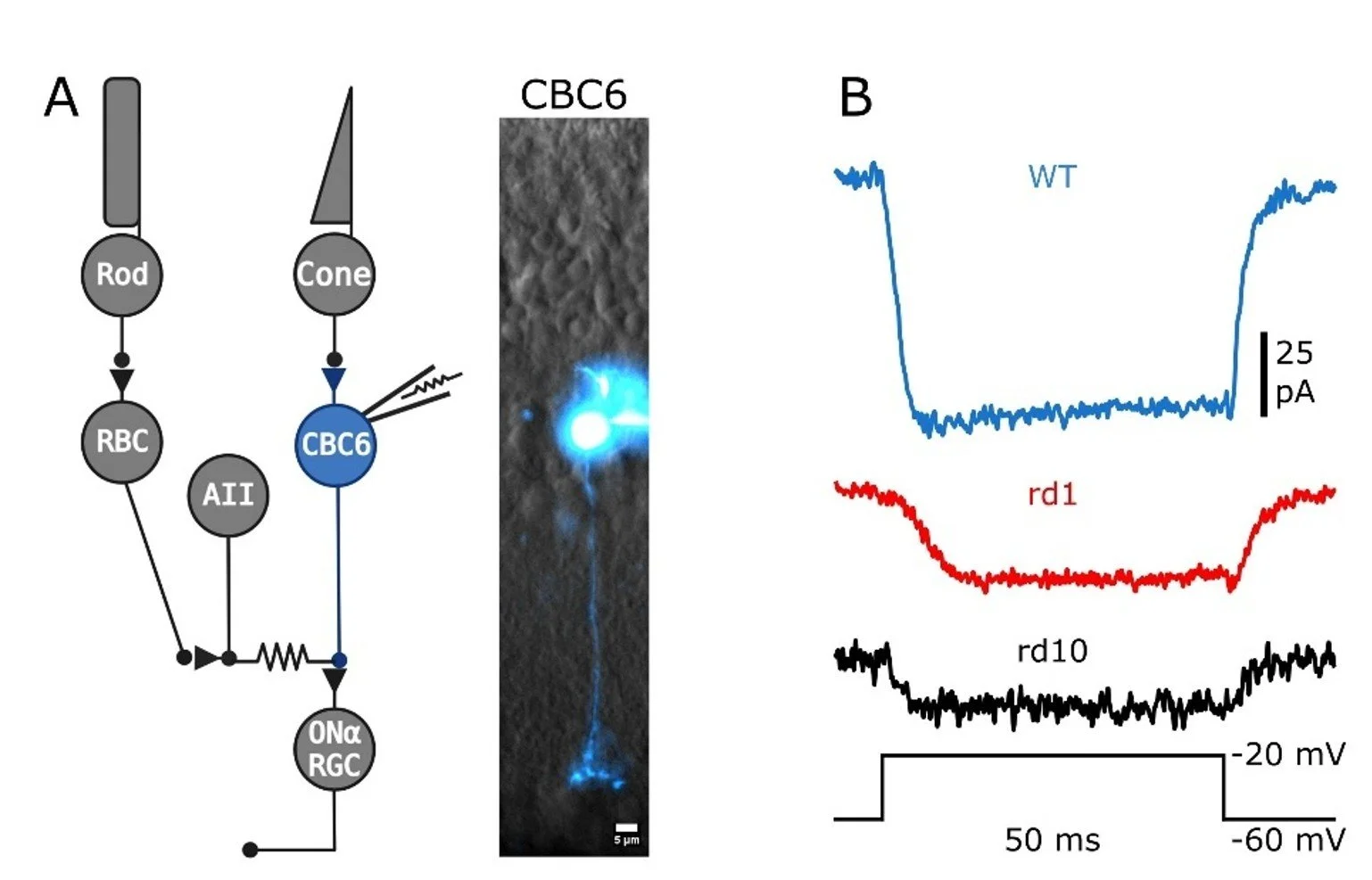
Patch-clamp electrophysiology in a retinal slice preparation. Here, a type-6 cone retinal bipolar cell has been patched with an electrode, allowing for high-resolution recording of ionic currents in the neuron. This bipolar cell receives visual signals from cone photoreceptors and relays them directly to retinal ganglion cells, which then transmit the information to the brain. (A). In response to a voltage step in membrane potential from -60 mV to -20 mV, voltage-gated calcium channels open generating inward current (B). In two mouse models of retinitis pigmentosa (rd1 and rd10), voltage-gated calcium current is reduced following photoreceptor degeneration.
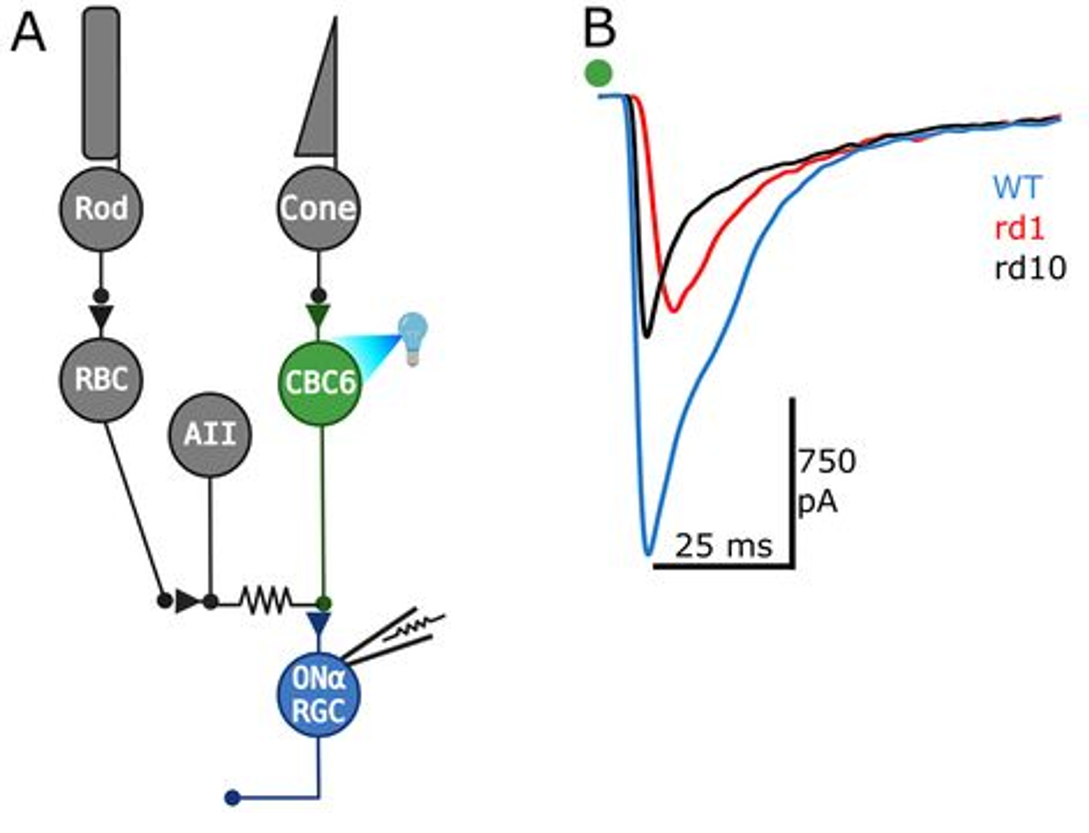
Optogenetic stimulation of 100’s of type-6 cone retinal bipolar cells in a flat-mount retina (A). The neurons express Channelrhodopsin-2, which is an ion channel that opens in response to blue light. Opening of the channel depolarizes the neurons, providing optical control over their activity. Excitatory post-synaptic currents (EPSCs) recorded in post-synaptic retinal ganglion cells reveal that neurotransmitter release is lowered in rd1 and rd10 models of retinitis pigmentosa.
Biological Preparations
Neurons for studying neuroscience

A flat-mount mouse retina. The retina is dissected as a cup, and four relieving cuts are made to create a clover shape. In this preparation, the retina is fixed in a chamber with a slice hold-down (harp), retinal ganglion cell side up for either patch-clamp electrophysiology or functional imaging (calcium imaging, etc).
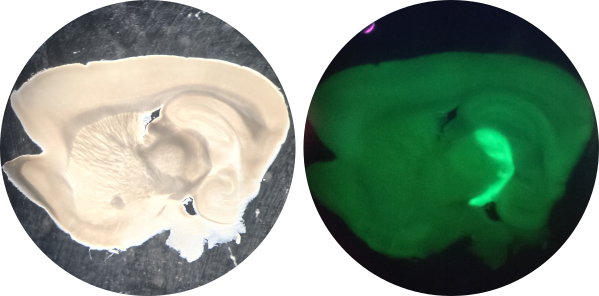
An acute parasagittal brain slice for electrophysiological and microscopy recording (Left). In this preparation, the brain is cut at a specific angle to preserve the axons from the optic nerve. A glutamate reporter (iGluSnFR) was virally delivered to retinal ganglion cells in the eye. The axons of the retinal ganglion cells (forming the optic nerve) are fluorescent, labeling the intact optic tract (Right).


An acute retinal slice preparation. The retina is a highly ordered tissue with many layers. Accessing cells in the middle of the retina can be difficult in the whole tissue. In this preparation, a flat retina is mounted on a piece of filter paper and cut in 250 micron increments with a Stoelting Tissue Slicer. The sliced paper with the retina is rotated 90 degrees and embedded in vacuum grease (Left), similar to cutting a sandwich in half and looking at the filling between the bread. This preparation provides physical access to all retinal neurons and plexiform layers (Right, transmitted IR Koehler Illumination with Dodt Gradient Contrast).
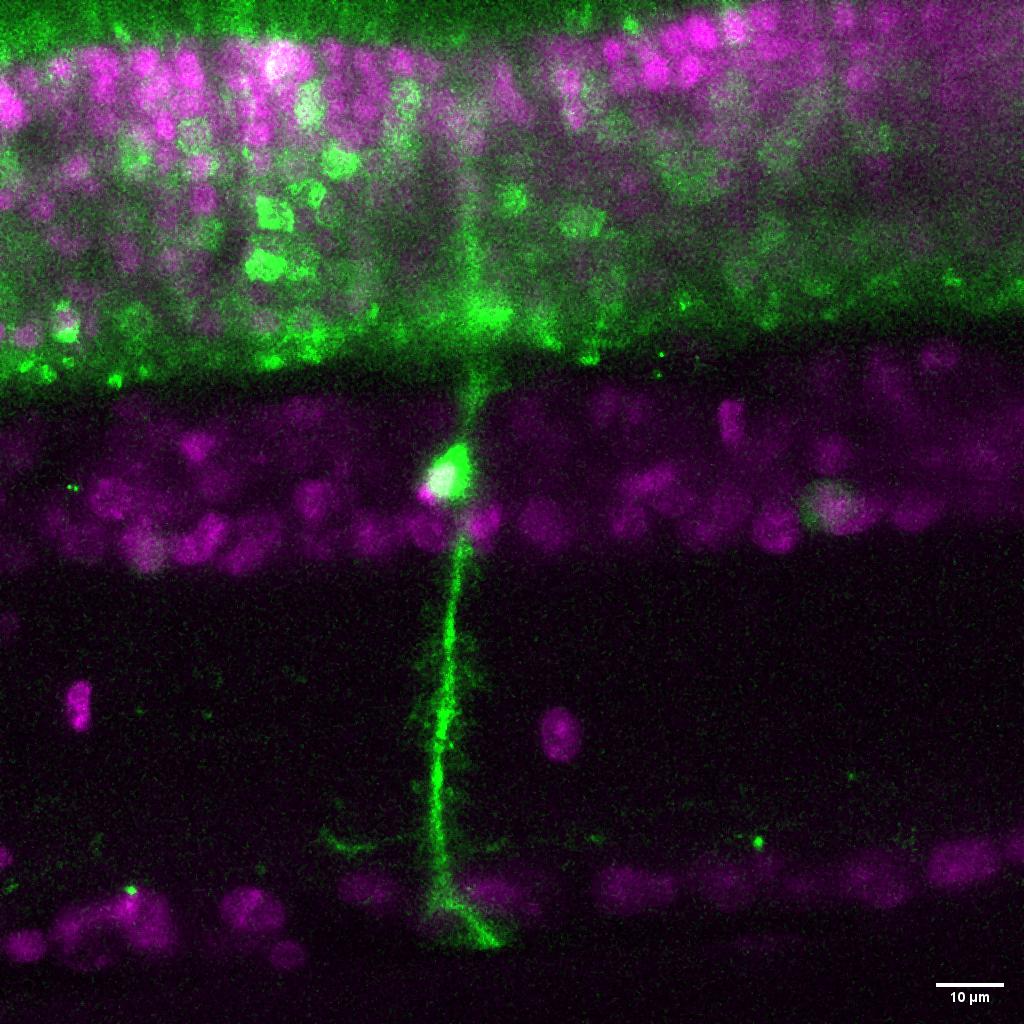
A Müller glia cell in a live retinal slice preparation. This glial cell has been virally transduced to express a construct containing a fluorescent protein. These glia play a critical role in maintaining retinal homeostasis and neuronal support.
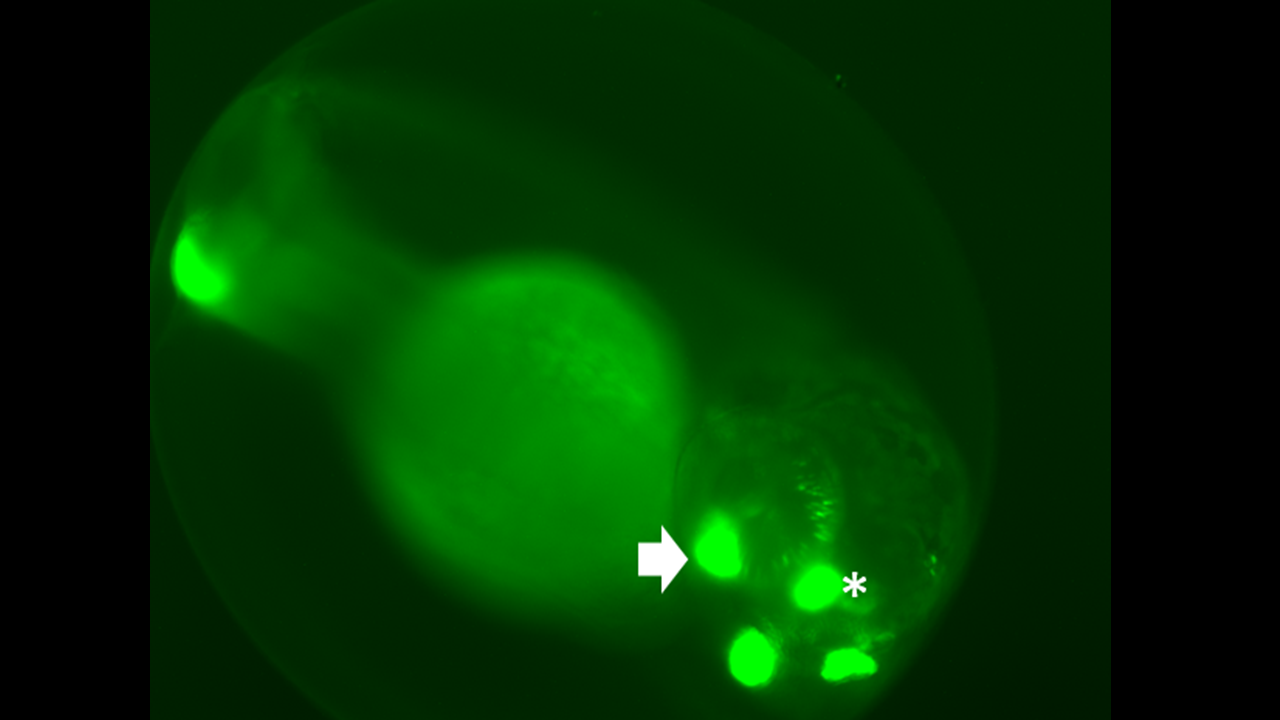
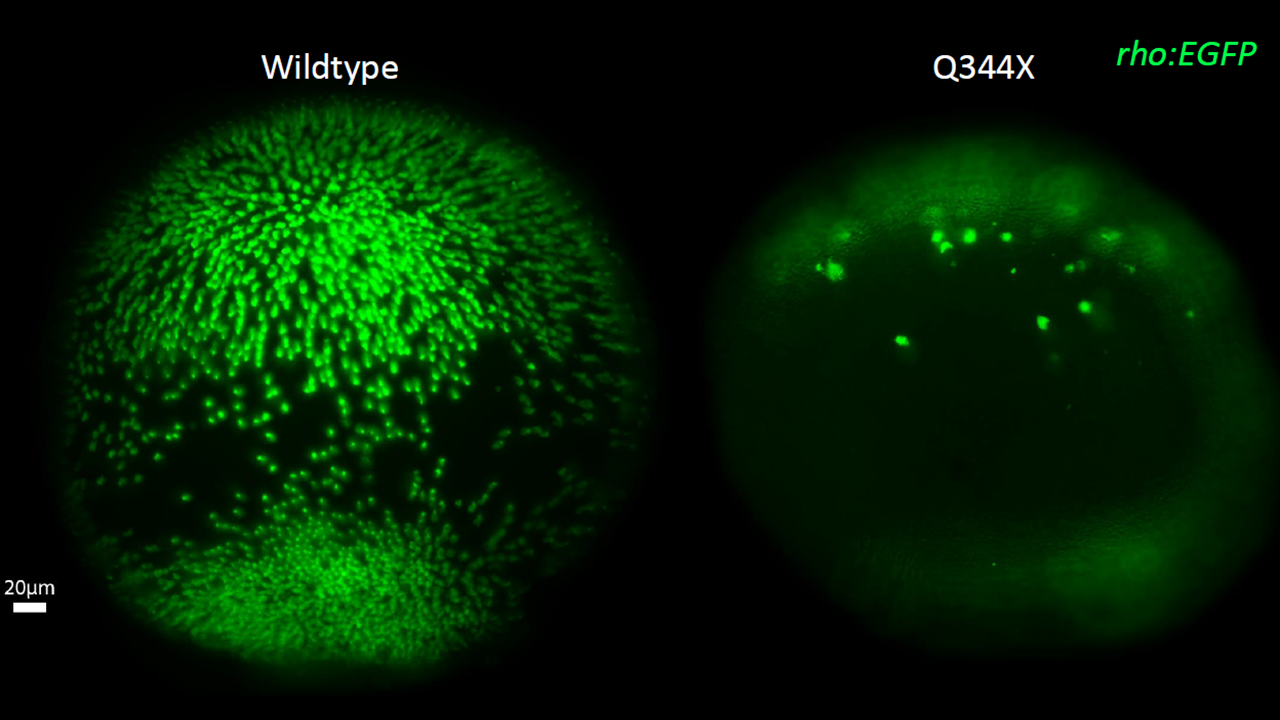
Zebrafish are a powerful tool for neuroscience research. This 3-day-old larva (Right) is expressing two transgenes: 1) a fluorescent green protein (GFP) under control of the rhodopsin promoter to label rod photoreceptors (asterisk) and 2) a transgene expressing a patient-derived mutant rhodopsin gene (Q344X) indicated by the expression of GFP in the olfactory bulbs (arrow). A whole-mounted wildtype zebrafish retina contains many rods at 7 days old (middle), while the Q344X mutant fish experiences severe rod degeneration (right). This rod death causes loss of scotopic vision and creates the basis for personalized medicine and drug discovery.
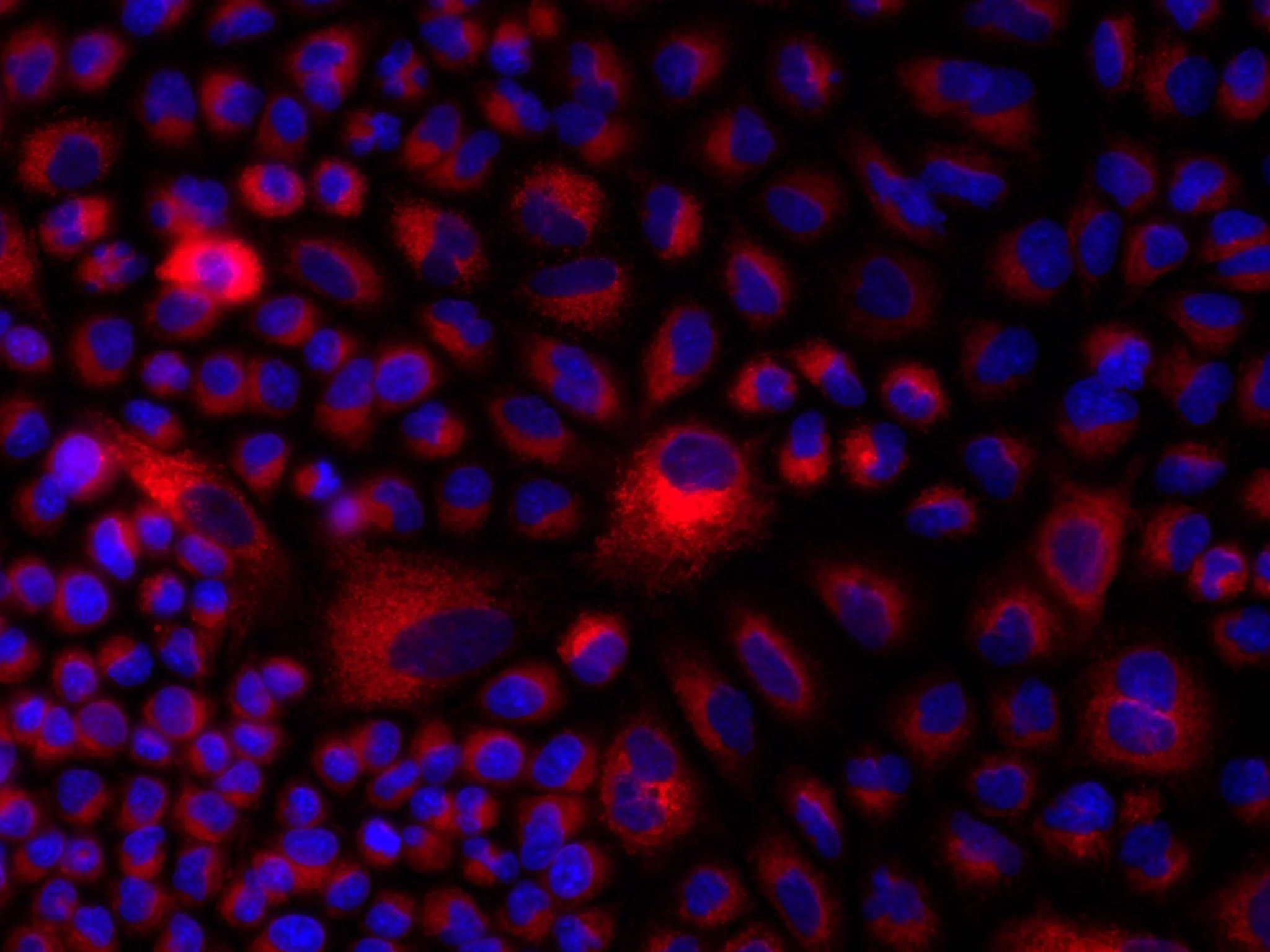
In vitro cell culture may not be a nervous system, but it allows for powerful high-throughput screening and pharmacological manipulation. In this image, cultured cells have taken up the Aldered dye to study the function of retinaldehyde dehydrogenase enzymes.